Discussion
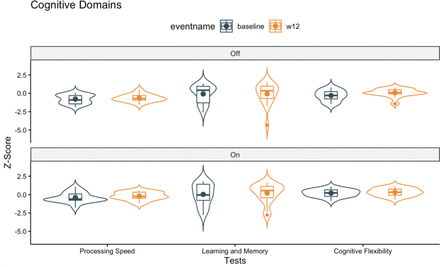
The current study used data from a randomised, sham-controlled, double-blind crossover trial to investigate the neuropsychological effects of active versus sham SCG-DBS over two 3-month periods in TRD. No significant differences in changes in depression or composite measures of cognitive domains were observed between active and sham SCG-DBS. Individual neuropsychological measures showed no deterioration. While Category Fluency Test performance, a measure of processing speed, improved after 3 months, this effect became non-significant after correcting for multiple comparisons and was not maintained after the crossover period. The absence of significant and sustained changes underscores the need for cautious interpretation and might reflect our small sample rather than the absence of true group differences.
Structural and functional brain alterations in TRD are associated with a higher risk of cognitive decline and its negative outcomes on functionality in this population. The SCG is a key target for DBS in treating TRD, primarily influencing mood through a top-down mechanism that enhances emotional regulation and alleviates depressive symptoms.10 SCG-DBS has demonstrated the ability to preserve cognitive functioning in TRD.6 12 19 In contrast, the ventral capsule (VC), another common target for DBS, operates via a bottom-up mechanism that directly activates motivational circuits and reward processing.20 Evidence indicates that while both targets aim to alleviate TRD symptoms, their cognitive impacts diverge significantly, with SCG stimulation being less likely to disrupt cognitive processes compared with VC stimulation.20 Consistent with our findings, studies that targeted the SCG in smaller samples found no deterioration in executive function, language, processing speed, attention or verbal fluency 12 months post-DBS surgery.13 19
The Category Fluency Test was the only assessment that demonstrated a short-term response to SCG-DBS at 3 months. However, this became non-significant after correcting for multiple comparisons and was not maintained after 6 months of stimulation. Research has shown that participants with MDD perform significantly worse than those with no psychiatric history on this test,21 reflecting impairments in processing speed. A small long-term study of open-label SCG-DBS with follow-up to 42 months reported individual variability in performance on the Category Fluency Test.14 As part of a network that also comprises cortical structures, stimulating the SCG via DBS may cause local and distributed effects.11 13 While category fluency is typically considered to index functions dependent on the lateral temporal and prefrontal cortex, tasks involving word generation or decisions to semantic cues have been shown to engage non-classical language areas such as the posterior cingulate cortex and adjacent precuneus.22 23 Functional neuroimaging studies of SCG-DBS demonstrate activation in this region with stimulation.24 25 Therefore, it is possible that DBS altered connectivity to such regions, although transiently, in our cohort.
Maintained cognitive functioning on the remaining tests in the domains of processing speed, learning and memory and cognitive flexibility is consistent with prior findings in TRD. Similar to our results, a previous trial of SCG-DBS in TRD reported stable performance on the Trail-making Test A at 1 year postsurgery and no univariate effect of time on the Trail-making Test B.19 Bogod et al14 also observed stable CVLT-II (Delayed Recall) performance in two participants throughout the study. Most of the extant literature indicates no significant change or, at most, a modest improvement in some cognitive measures.14 19 A recent meta-analysis suggests that it may take more than 6 months of DBS treatment for significant cognitive gains.9 While our study was unable to assess the long-term effects of SCG-DBS, supporting evidence also suggests that it may take time for improvements to emerge.26 Cognitive improvements reported in some open-label studies6 7 may reflect practice (ie, due to repeated administration of neuropsychological tests) or placebo effects, which are minimised in our crossover sham-controlled design. Moreover, the cohort for our study also performed relatively well at baseline, despite selecting measures from domains previously demonstrated to be impaired in MDD.1 In our study, only one test score (ie, GPT) was more than one SD below the normative mean value. Thus, the opportunity to observe ‘normalisation’ on a statistically reliable basis may have been blunted. Future studies should enrol participants with considerable cognitive deficits in addition to TRD to better assess the effects of SCG-DBS on cognitive function.
Moreover, no significant changes in HDRS-17 scores were observed in this study. Cognitive improvements in individuals with MDD are often linked to reductions in depressive symptoms. A systematic review and meta-analysis of 69 studies found that both symptom improvement and the number of prior depressive episodes influenced cognitive changes.27 Thus, the lack of significant changes in depressive symptoms in our study may have contributed to the absence of cognitive improvement following SCG-DBS. Moreover, the SCG, a key component of the mood-regulating circuit, shows reduced activity following antidepressant treatments and is the primary target for DBS.28 However, SCG-DBS trials for TRD have yielded inconsistent results regarding efficacy and sustainability. While some studies report significant improvements in depressive symptoms following SCG-DBS,11 29 a 6-month double-blind, sham-controlled trial found no statistically significant antidepressant effects despite demonstrating the safety and feasibility of SCG-DBS.30Additionally, our study demonstrated non-significant changes in depressive symptoms following SCG-DBS, highlighting the need to consider potential placebo effects. Furthermore, the antidepressant effects of DBS may develop in a delayed and progressive manner, with differences between active and sham stimulation potentially not evident until after 1–2 years of treatment, posing significant challenges for sham-controlled studies.
A strength of this study is that the randomised crossover design combined with the double-blinding enhances the robustness. Additionally, employing both active and sham treatment groups mitigated confounding effects arising from repeated neuropsychological testing, such as practice and task familiarity. However, some limitations warrant consideration. The potential for carryover effects, particularly in the absence of a sufficient washout period, remains an important methodological concern. Similarly, the impact of concomitant antidepressant treatments on cognitive performance was not assessed, and the 4-week stable antidepressant regimen prior to the DBS surgery might not have been sufficient to stabilise clinical changes fully. Another limitation lies in the use of two distinct medical devices (ie, St Jude Medical/Abbott Libra or Medtronic Activa), which could have complicated the results. Furthermore, participants were not matched across groups based on their treatment history, concurrent treatment regimens or depressive symptoms. While including these covariates in MMRMs might have increased model complexity, baseline HDRS-17 scores did not differ significantly between groups, suggesting a limited impact on findings. Given the study’s small sample size and the primary aim of detecting differences, the results should be interpreted with caution, as insufficient statistical power may have obscured subtle group differences. Nonetheless, these results contribute valuable insights to the growing body of literature on SCG-DBS and its cognitive effects. Moreover, the relatively short stimulation period might have prevented clinical benefits from emerging. Future studies should enrol larger samples and implement longer stimulation periods to provide a more comprehensive evaluation of SCG-DBS and its effects.