Methods
Study design
We conducted an observational longitudinal cohort study to investigate SGA and metformin co-prescribing in UK primary care from 1 January 2005 to 31 December 2019.
Data source
We used data from the Clinical Practice Research Datalink (CPRD), which includes two anonymised databases: GOLD26 and Aurum.27 These databases include current and historical primary care records for >62 million patients. We used the April 2023 and May 2022 database builds of GOLD and Aurum, respectively. For patients registered at primary care practices in England, CPRD data are linked to hospitalisation records (Hospital Episode Statistics) and to area-level deprivation data (the 2019 English Index of Multiple Deprivation (IMD)).
Population
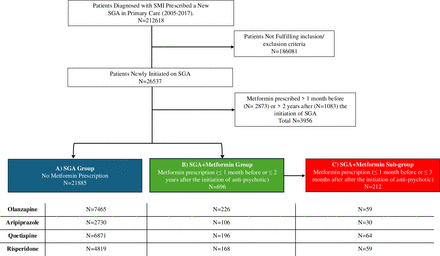
The study cohort included patients newly prescribed aripiprazole, olanzapine, quetiapine or risperidone in primary care—the four most frequently prescribed antipsychotics in the UK4—and has been previously characterised.28 Inclusion criteria, applied as at the date of first prescription for the SGA, were: age 18–99 years; SMI diagnosis recorded in primary care (ie, schizophrenia, bipolar disorder or other non-organic psychoses (eg, schizoaffective disorders, delusional disorder, psychotic episodes, non-organic psychosis not otherwise specified)); primary care practice registration for at least 6 months and at least one lipid or glycated haemoglobin (HbA1c) measurement recorded in the last 2 years. Exclusion criteria were: prescription of more than one antipsychotic (or a long-acting injectable antipsychotic in the prior 90 days) and dementia diagnosis.
Patients meeting eligibility criteria entered the study on their first prescription (index) date between 1 January 2005 and 31 December 2017 and completed the study at the earliest of: completion of 2-year follow-up (final follow-up, 31 December 2019), end of primary care registration, death or the last data collection date from the primary care practice. Further cohort details and other outcomes are reported in the study by Richards-Belle et al.28
Data management
To identify antipsychotic4 and metformin (online supplemental table 1) prescriptions, we developed search strategies (which considered generic and brand names) to identify relevant product codes in CPRD product code dictionaries. We used the resulting code lists to extract data on these medications from patient prescription records.
To characterise the cohort, we extracted data on the following demographic and baseline characteristics: sex, ethnicity, SMI diagnosis, age (at first SMI diagnosis, first antipsychotic prescription and at index date), geographical region, area-level deprivation, comorbidities (cerebrovascular disease, dyslipidaemia, myocardial infarction, hypertension, liver disease, renal disease, diabetes, alcohol misuse, substance misuse, polycystic ovary syndrome), concomitant medication prescriptions (antidepressants, lipid-regulating medications, insulin), biochemical parameters (HbA1c, random glucose), smoking status, body mass index (BMI) and body weight.
For patients in England, if ethnicity was not coded in the primary care record, we supplemented it with ethnicity data from linked HES records, where available. If a patient had more than one ethnicity category recorded, we used the most frequent (or the most recent, if frequencies were equal). Geographic region refers to the patient’s registered primary care practice and includes Northern Ireland, Scotland, Wales and nine regions across England as per the Office for National Statistics categories. For patients in England, relative deprivation was derived from linked small area-level data using the 2019 English IMD, based on either the patient’s residential postcode or, if unavailable, the primary care practice postcode. If a patient had multiple SMI diagnoses recorded, then the most recent diagnosis category as at the index date was used, retaining the first diagnosis date. Binary indicators were used to define antidepressant prescriptions (as categorised by the British National Formulary) and insulin prescriptions in the 2 years prior to the index date. BMI (kg/m2) was categorised as underweight (<18.5), healthy weight (≥18.5 to <25), overweight (≥25 to <30) and obese (≥30).
Missing data
Missing weight values and covariates were handled using multiple imputation by chained equations using the ‘mice’ package in R. The imputation model included baseline, outcome and auxiliary variables and we assumed data were missing at random. We generated 25 imputed datasets and pooled results across imputed datasets according to Rubin’s rules.
Statistical analysis
All analyses were conducted in R (V.4.4.1). The full analytical code is available at: https://github.com/Alvin-RB/antipsychotic_metformin_coprescription.
To determine the incidence and prevalence of metformin prescription (objective 1), we first calculated the number of patients newly prescribed metformin in the following time-windows: ≥1 month before the incident SGA, ≤1 month before to ≤2 years after incident SGA, ≥2 years after the incident SGA. We then estimated the cumulative incidence of metformin co-prescription, using the Kaplan-Meier method to model the time to first metformin prescription among patients not previously prescribed metformin >1 month prior to the index date. To ensure that patients who were prescribed metformin on, or within 1 month prior to, the index date were included, we set their follow-up time at 0.5 days. For all other patients, follow-up time was censored at the earliest date of: first metformin prescription, death, end of primary care registration, last data collection from the primary care practice or completion of 2 years follow-up from index date. To examine prescribing prevalence over time, we calculated annual prevalences (standardised per 1000 patients). The numerator was the total number of unique patients with at least one metformin prescription each year from 2005 to 2017, and the denominator was the total number of eligible patients alive and remaining in follow-up during the given year.
To compare the demographic and baseline characteristics of patients who were prescribed metformin and those who were not (objective 2), we stratified descriptive statistics of the two groups by metformin exposure status. The first group comprised patients who had never been prescribed metformin, referred to as the SGA-only group and the second group comprised patients first prescribed metformin ≤1 month prior to or ≤2 years after the initiation of the SGA, referred to as the SGA+metformin group. Similarly, to investigate the potential rationale for prescribing, we compared the characteristics of patients in the SGA+metformin group with and without a recorded potential indication for metformin (ie, diabetes and/or PCOS).
To describe changes in weight over time among patients prescribed and not prescribed metformin (objective 3), we plotted descriptive line graphs of the mean weight at baseline and at 6-month, 1-year and 2-year follow-up, as well as the percentage change from baseline at each follow-up time-point (all with 95% CIs). Among patients in the SGA+metformin group, we included only those who were prescribed metformin ≤1 month prior to or ≤3 months after the index date (ie, those first prescribed metformin in close proximity to SGA initiation). This time range was selected because the first outcome time-point was 6 months, and we hypothesised that metformin initiated >3 months prior to the outcome would not allow sufficient time for the medication to exert an effect. In these primary analyses, we handled missing values using multiple imputation (see ‘Missing data’ section), but we also explored the impact of missing data through an observed-values analysis.
To estimate the potential effect of metformin on weight change, we constructed linear regression models to estimate the mean difference in weight (kg) at 6 months, 1 year and 2 years postindex date. The models included an indicator for metformin status as the main exposure and weight (kg) as the outcome. In an initial model (model 1), we adjusted for the baseline weight. In model 2, we adjusted for all the confounders identified using a directed acyclic graph (online supplemental figure 2), created to determine confounders of the relationship between metformin prescription and weight change. The variables included in the models were baseline weight (kg), antipsychotic medication, sex, age at index date, ethnicity, social deprivation, prior diagnosis of diabetes and prior diagnosis of PCOS. Models were stratified by sex, as PCOS—a key confounder—only affects females. The models were parameterised with linear terms for all variables and additional quadratic terms for continuous variables. As mentioned above, among patients in the SGA+metformin group, we included only those prescribed metformin ≤1 month prior to or ≤3 months after the index date.